In the dyeing and printing industry, bleaching with bleaching agents and whitening with brighteners are two important methods for obtaining white polyester fabrics.
Among them, whitening with brighteners has the advantages of low brightener dosage, good whitening effect and low fiber damage. Even bleached fabrics usually need to be further whitened with brighteners. Stilbene (ER) type fluorescent brighteners are an important type of brightener used for polyester fabrics. When commercialized, manufacturers often use grinding and dispersion methods to make them into liquid products. At present, poor dispersion stability is an important problem affecting the development of liquid fluorescent brighteners. Many related products are prone to solid particle precipitation and sedimentation after being placed for several weeks or months, resulting in poor whitening effect on polyester fabrics, and even causing uneven whitening, color spots, color spots and other defects in the fabric samples. Therefore, research and development of highly stable liquid fluorescent brighteners has important practical value. This paper takes the fluorescent whitening agent ER-I as an example, and uses a grinding method to prepare it into a liquid fluorescent whitening agent. The effects of factors such as whitening agent particle size, dispersant type, whitening agent to dispersant mass ratio, and whitening agent concentration on the stability of the dispersion system were investigated. An orthogonal experiment was designed to determine the degree of influence of each factor, aiming to provide a useful reference for the development of highly stable liquid fluorescent whitening agents.
1. Experimental Section
1.1 Materials, Reagents, and Instruments
Materials: Polyester knitted fabric (230 g/m²)
Reagents: Whitening agent ER-I, defoamer, nonionic dispersant (polyoxyethylene ether type), anionic dispersant MF, and superdispersant (mainly a copolymer based on acrylic acid and styrene, Mw 1.49 × 10⁵) were all commercially available. Instruments: 756S UV-Vis spectrophotometer (Shanghai Lingguang Technology Co., Ltd.), RTSM-0.5BJD horizontal sand mill (Shanghai Rute Electromechanical Equipment Co., Ltd.), 800B low-speed centrifuge (Shanghai Anting Scientific Instrument Factory), Nano ZS90 nanoparticle size and Zeta potential analyzer (Malvern Instruments, UK), Mars40 rheometer (HAAKE, Germany), DYE-24 staining machine (Shanghai Qianli Automation Equipment Co., Ltd.), WSD-3C whiteness meter (Beijing Kangguang Optical Instrument Co., Ltd.).
1.2 Preparation steps of liquid fluorescent whitening agent: Whitening agent, dispersant, zirconium beads, and water were added sequentially to a laboratory micro-grinding device. The grinding speed was adjusted and timed. If foaming occurred, an appropriate amount of defoamer could be added. During the grinding process, the average particle size of the whitening agent was measured and analyzed using a nanoparticle size analyzer. After grinding, the zirconium beads were removed by filtration through gauze. The resulting filtrate was then diluted to a final volume to obtain the liquid fluorescent whitening agent.
1.3 Centrifugation Stability Test Procedure
A suitable amount of the liquid fluorescent whitening agent was placed in a centrifuge tube. After centrifugation at 2000 r/min for a specific time, 1 mL of the intermediate layer liquid after centrifugation and 1 mL of the liquid before centrifugation were taken. These were diluted with an equal volume of N,N-dimethylformamide, and the absorbance at the maximum absorption wavelength was measured using a UV-Vis spectrophotometer. The residual rate was calculated using the following formula:
Residual Rate/% = (A<sub>after</sub> / A<sub>before</sub>) × 100%
Where A<sub>after</sub> and A<sub>before</sub> are the absorbance values of the dispersions before and after centrifugation, respectively. 1.4 Polyester Whitening Process
Prepare the dye liquor according to the whitening agent dosage x% owf and liquor ratio 1:30. Place the polyester fabric in the liquor, set the initial dyeing temperature of the dyeing machine to 40℃, increase the temperature to 130℃ at a rate of 2℃/min, and hold for 60 min. Then cool to 40℃, remove the fabric, wash with water, and air dry.
1.5 Whiteness and Color Parameter Testing Methods
Use a whiteness meter to test the whiteness value of the whitened samples. The average of three tests for each sample is taken as the final whiteness value.
Color parameters (L, a, and b) are also read from the whiteness meter. 3
2. Results and Discussion
2.1 Single Factor Analysis
2.1.1 Effect of Particle Size of Brightener on Stability
The brightener ER-I was ground using a nonionic dispersant. The average particle size of the particles in the obtained brightener dispersion was adjusted by controlling the grinding time. Dispersions with average particle sizes of 260 nm, 400 nm, 630 nm and 1080 nm were obtained [9]. Their centrifugal stability was tested, and the results are shown in Figure 1. As can be seen from the figure, the residual rate of brightener in the dispersion decreased rapidly within 15 min of centrifugation and then tended to level off. The longer the centrifugation time, the more severe the sedimentation of brightener particles and the lower the residual amount of brightener in the dispersion. Under the same centrifugation time conditions, the larger the particle size of brightener particles, the lower the residual rate of the dispersion. This indicates that the dispersion system with large particle size has a weaker ability to maintain stability. Therefore, a sufficiently small particle size is a necessary condition for preparing stable liquid fluorescent brighteners.
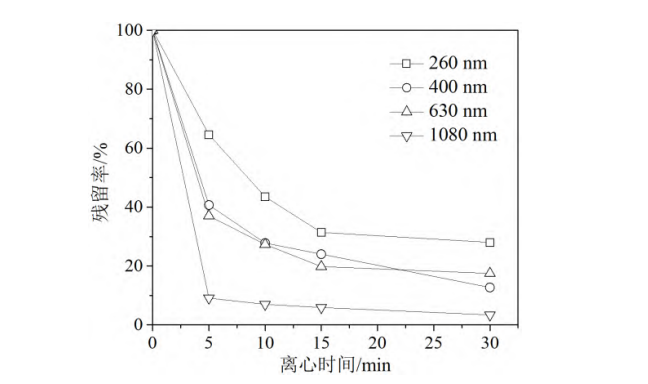
Grinding conditions: nonionic dispersant, m(brightener)/m(dispersant) 2/1, brightener concentration 1 g/L
Figure 1 Variation curves of whitening agent residual rate with centrifugation time under different particle size conditions
2.1.2 Effect of the Mass Ratio of Brightener to Dispersant on Stability
During the grinding process, the amount of brightener was kept constant, and the brightener was ground until the average particle size reached 200 nm (due to the large error in the particle size control experiment, this paper uses the particle size level to express the particle size range). The effect of the mass ratio of brightener to dispersant on the centrifugal stability of the dispersion system was investigated by changing the amount of nonionic dispersant. The brightener residue rate after centrifugation for 15 min under different dispersant dosages is shown in Figure 2. As can be seen from the figure, when the mass ratio of brightener to dispersant increased from 1:0.5 to 1:0.7, the brightener residue rate in the dispersion increased from 31.3% to 47.8%. Further increasing the dispersant dosage resulted in a decrease in residue rate. This result indicates that the amount of dispersant used needs to be maintained at an appropriate level; too much or too little is not conducive to obtaining high stability.
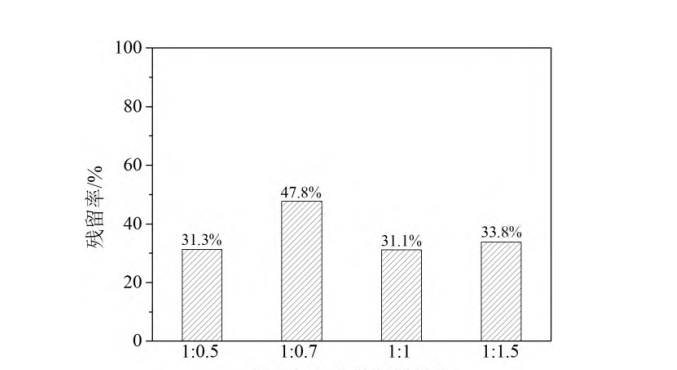
mass ratio of whitening agent to dispersant
Figure 2 Residual rate of whitening agent in centrifuged dispersion under different dispersant dosage conditions
2.1.3 Effect of Brightening Agent Concentration on Centrifugal Stability
The brightening agent was ground using a nonionic surfactant to obtain dispersions with different brightening agent concentrations. The effect of brightening agent concentration on the centrifugal stability of the dispersions was investigated, and the results are shown in Figure 3. As shown in the figure, as the brightening agent concentration in the dispersion increased from 1 g/L to 15 g/L, the residual rate of the brightening agent gradually decreased from 31.4% to 15.1%. This indicates that with the increase of brightening agent concentration, the average distance between brightening agent particles decreases, making the particles more prone to aggregation and sedimentation.
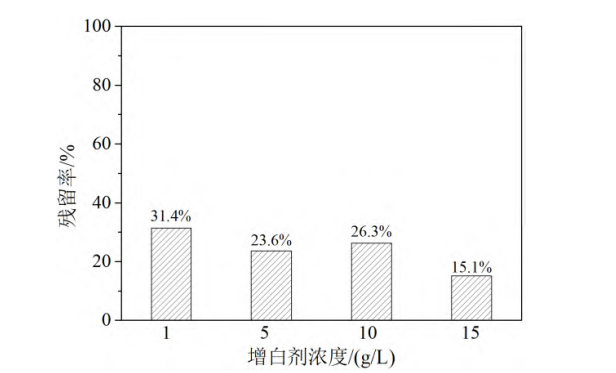
Milling conditions: nonionic dispersant, m(brightener)/m(dispersant) 2/1, particle size level 200 nm
Figure 3 Residual rate of whitening agent in centrifuged dispersion under different whitening agent concentration conditions
2.1.4 Effect of Dispersant Type on Stability Anionic, nonionic, compound (anionic and nonionic in a 1:1 mass ratio), and superdispersant were used to grind the whitening agent under the same conditions. The centrifugal stability of the resulting dispersions was tested to explore the effect of dispersant type on stability. The results are shown in Figure 4. As shown in the figure, the dispersion obtained using nonionic dispersant under the same conditions had the lowest residual rate, only 34.9%. The residual rate increased significantly when using anionic, compound, or superdispersant dispersants, reaching 47.4%–53.1%. Among them, the centrifugal residual rate was the highest when using superdispersant, reaching 53.1%. This may be because the anionic groups contained in these dispersants significantly increased the negative charge repulsion between particles, achieving a stabilizing effect.
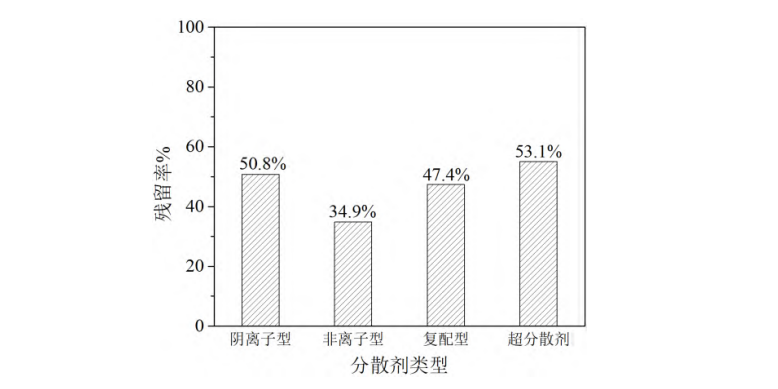
Milling conditions: m(brightener)/m(dispersant) 2/1, brightener concentration 1 g/L, particle size 200 nm
Figure 4 Residual rate of whitening agent in centrifuged dispersion using different dispersants
2.2 Orthogonal Experimental Design and Result Analysis The aforementioned research results show that the average particle size of the whitening agent, the type of dispersant, the amount of dispersant, and the concentration of the whitening agent all have a significant impact on the stability of the whitening agent dispersion. To further study the influence of each parameter on the stability of the liquid whitening agent, based on the single-factor experiment, an L16 (44) orthogonal experimental design was carried out with factors such as average particle size level (A), whitening agent concentration (B), whitening agent to dispersant mass ratio (C), and dispersant type (D). The orthogonal experimental table is shown in Table 1.
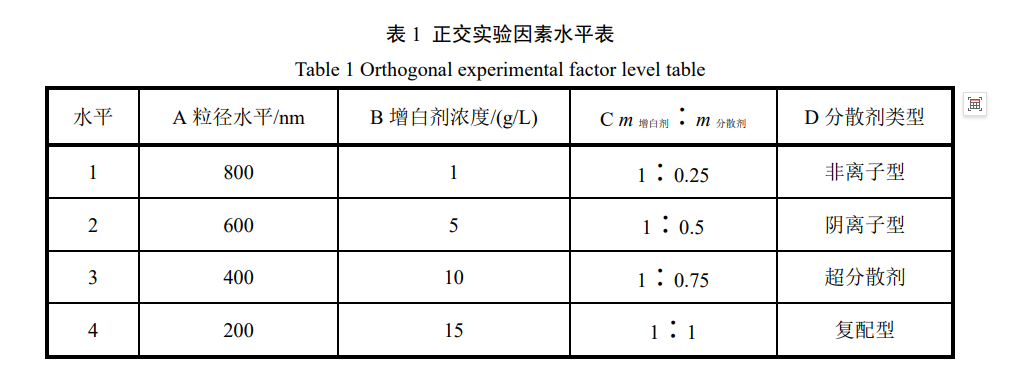
Using the residual amount of whitening agent in the dispersion after centrifugation as an indicator, the optimal grinding process conditions for the whitening agent were determined, and the orthogonal experimental results are shown in Table 2. Based on the experimental data in Table 2, range analysis was used to determine the degree of influence of each factor. Table 2 shows that factor A has the largest range R value, reaching 33.30. This indicates that the average particle size of the whitening agent has the greatest impact on the stability of the dispersion. Specifically, when the average particle size is at the 200 nm level, the centrifugation residue rate of the resulting dispersion is the highest.
The range values of factors B to D are 10.40, 12.85, and 18.53, respectively. Therefore, the order of influence of each factor is: average particle size of whitening agent > type of dispersant > mass ratio of whitening agent to dispersant > concentration of whitening agent. Based on the numerical values of each level, the optimal combination is A4B1C2D3. That is, when the concentration of the fluorescent whitening agent is 1 g/L, a superdispersant is used, the mass ratio of whitening agent to dispersant is set at 1:0.5, and the average particle size of the whitening agent is ground to the 200 nm level, the best stability effect should be obtained (this condition happens to be consistent with the conditions in Section 2.1.4 when using a superdispersant). Further verification showed that the whitening agent dispersion obtained under the above optimal conditions achieved a residue rate of 59.5% after centrifugation.
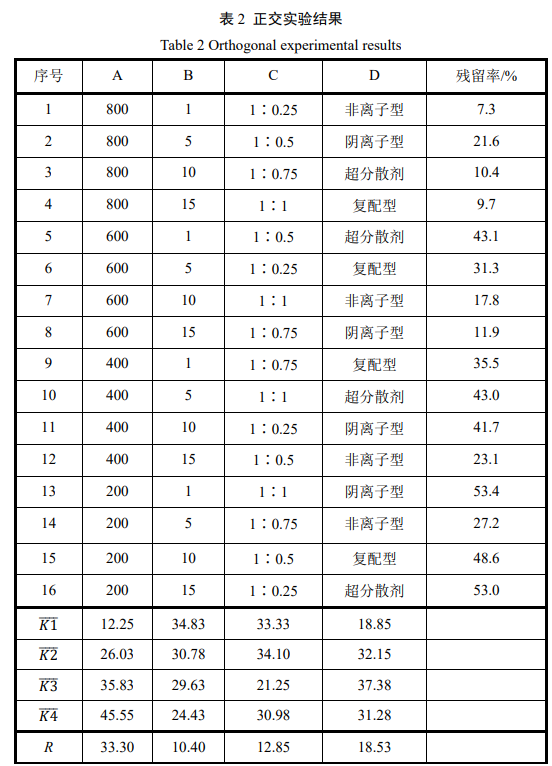
2.3 Static Stability of Optimized Samples
The liquid fluorescent whitening agent obtained under optimized conditions was allowed to stand at room temperature (25±5℃) for a period of time. Sedimentation was observed, and the residual amount of whitening agent in the solution was tested. The results are shown in Figure 5. As shown in Figure 5, the optimized sample exhibited better sedimentation resistance compared to the control sample. After 74 days, a significant concentration difference was observed between the upper and lower layers of the control sample. Testing revealed that the residual whitening agent in the control sample solution was only 11.7%, while the residual whitening agent in the optimized sample solution was still 50.5%. This result indicates that the optimized sample based on the superdispersant has significantly better static stability than the nonionic dispersion system.
Note: The optimized sample is the sample prepared using orthogonal experimental optimization conditions; the control sample is the sample prepared using a nonionic dispersant, with other preparation conditions being the same as the optimized sample.
Figure 5 The variation curves of whitening agent residual rate with settling time for optimized and compared samples
2.4 Stability Mechanism Analysis of the Hyperdispersant System
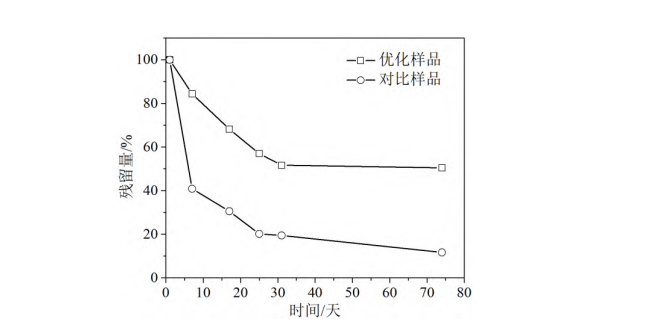
To reveal the reason for the higher stability of the optimized sample based on the hyperdispersant, the Zeta potentials of the optimized sample and the control sample were measured, and the results are shown in Table 3. Table 3 shows that the Zeta potential of the optimized sample is -38.2 eV, which is almost twice that of the control sample. This result reflects that the hyperdispersant system with numerous anionic groups can effectively provide repulsive forces between the whitening agent micelles, thereby reducing the probability of whitening agent molecule aggregation and sedimentation, and making the dispersion system more stable.
As shown in Figure 6, the viscosity variation curves with shear rate indicate that the optimized sample exhibits rheological properties that are drastically different from those of the control sample. The control sample prepared with a nonionic dispersant exhibits shear thickening, meaning its viscosity gradually increases with increasing shear rate. In contrast, the optimized sample prepared with a superdispersant exhibits shear-thinning fluid behavior, meaning its viscosity gradually decreases with increasing shear rate [10]. Overall, the optimized sample has a higher viscosity value than the control sample.
It is inferred that in the optimized sample system, the superdispersant and the whitening agent particles are firmly adsorbed through anchoring, thus forming relatively stable whitening agent micelles. The solvated long chains of the superdispersant provide bridging for the formation of a three-dimensional network structure between the micelles and provide a large energy barrier to suppress van der Waals attraction between micelles, thereby enabling the dispersion system to reach a thermodynamically stable state.
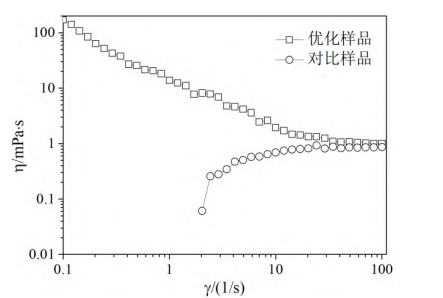
Figure 6 Variation curves of viscosity with shear rate of optimized and compared samples
2.5 Whitening Effect of Optimized Samples
The optimized sample and the control sample were used to whiten polyester fabrics using a high-temperature, high-pressure impregnation method. The whiteness and color parameters of the whitened polyester were tested, and the results are shown in Table 4. Compared with the original polyester sample, the whiteness of the fabric was significantly increased after treatment with the whitening agent. Within the yellowing point of the whitening agent, the whiteness value of the polyester fabric also increased significantly with the increase of the whitening agent dosage. When the whitening agent dosage was 0.2% owf, the whiteness value of the whitened fabric of the optimized sample reached 93.74. Compared with the control sample, the optimized sample showed higher whiteness at the same whitening agent dosage. This indicates that the optimized dispersion system in this paper has a significant promoting effect on the adsorption and penetration of whitening agent molecules into polyester fibers.
3. Conclusion
The factors affecting the stability of optical brighteners are as follows: particle size > type of dispersant > mass ratio of optical brightener to dispersant > concentration of optical brightener. The optimal conditions for obtaining a highly stable liquid optical brightener are: using a superdispersant, with an optical brightener concentration of 1 g/L and an optical brightener to dispersant mass ratio of 1:0.5, grinding the optical brightener particles to an average particle size of approximately 200 nm.
Under the optimized conditions, the centrifugal residue rate of the optical brightener dispersion system was 59.5%, and after standing for 74 days, the optical brightener residue rate in the dispersion was 50.5%, both results being significantly better than the comparative sample. Furthermore, the optimized liquid optical brightener obtained in this paper exhibits good whitening effects on polyester fabrics.

 EN
EN 中文
中文 ES
ES




.jpg)













